From development to production.
A single source solution.
Transfer from Design Controls to a validated medical device manufacturing and packaging process ensures that continuity is maintained as the product is commercialized. This vertical integration helps reduce costs, mitigate risk, streamline supply chain, and ensure consistent quality.
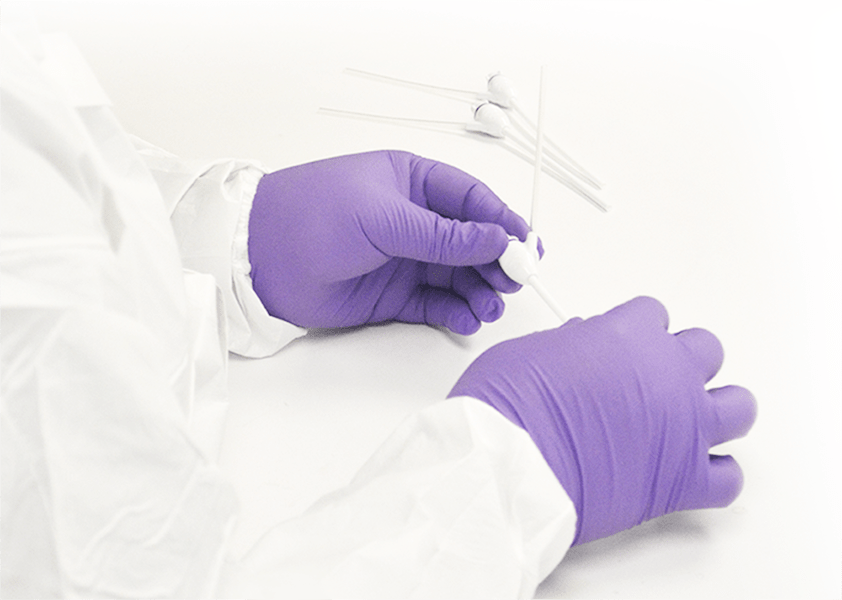
Design Verification and Validation Builds
Component manufacturing, assembling and packaging all built with complete batch traceability and quality controls for various validation activities like sterilization, biocompatibility and packaging





